 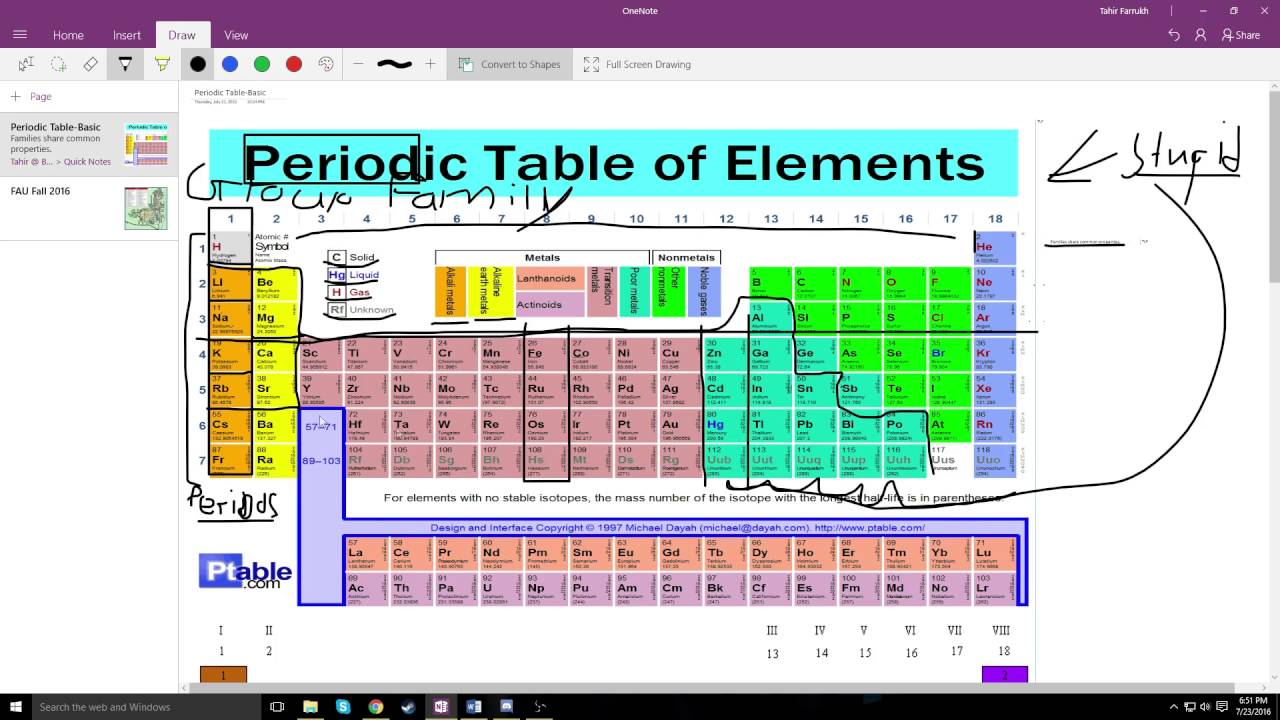
Based on the group valency of elements write the molecular formula of the following compounds giving justification for each:(i) Oxide of first group elements.(ii) Halide of the elements of group thirteen, and(iii) Compound formed when an element, A of group 2 combines with an element, B of group seventeen.The key difference between periods and groups is that the periods are horizontal rows whereas the groups are the vertical columns in the periodic table of chemical elements.What is the atomic number of element of period 3 and group 17 of the Periodic Table?1041721.Atomic number of a few elements are given below 10, 20, 7, 14(a) Identify the elements(b) Identify the Group number of these elements in the Periodic Table(c) Identify the Periods of these elements in the Periodic Table(d) What would be the electronic configuration for each of these elements?(e) Determine the valency of these elements.Its position in modern periodic table will be(a) Group 1 and Period 3 (b) Group 2 and Period 3(c) Group 13 and Period 3 (d) Group 16 and Period 3 An element ‘X’ is forming an acidic oxide.Which of the following statement (s) about the Modern Periodic Table are incorrectThe elements in the Modern Periodic Table are arranged based on their decreasing atomic numberThe elements in the Modern Periodic Table are arranged based on their increasing atomic massesIsotopes are placed in adjoining group (s) in the Periodic TableThe elements in the Modern Periodic Table are arranged based on their increasing atomic number(a) (i) only(b) (i), (ii) and (iii)(c) (i), (ii) and (iv) (d) (iv) only. 
Give examples of alkaline metals present in Group-1 in the Periodic Table.(f) According to Newlands’ classification of elements, the properties of sulphur are similar to those of oxygen because sulphur is the _ element starting from oxygen. (b) The horizontal rows in a periodic table are called _. Fill in the following blanks with suitable words:(a) The basis for modern periodic table is _.How does the valency of elements vary in going down a group of the periodic table?. 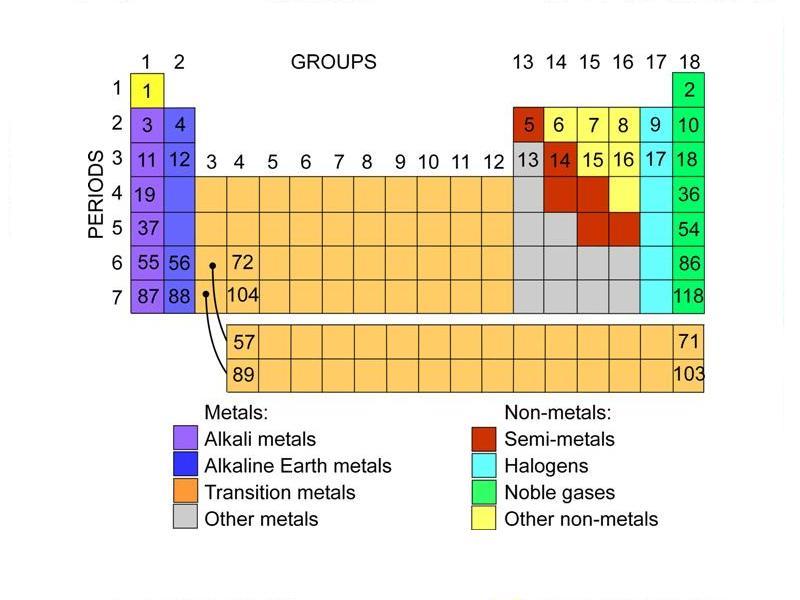
In which group of Mendeleev's periodic table is this element placed?(a) group II (b) group III (c) group V (d) group VIII Its most probable position in the modern periodic table is(A) Group 1 and Period 3 (B) Group 16 and Period 3 (C) Group 17 and Period 3 (D) Group 2 and Period 3
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
